PHYSICAL PARAMETERS OF SYNTHESIZED HETEROMETALLIC COMPLEX COMPOUND
Abstract
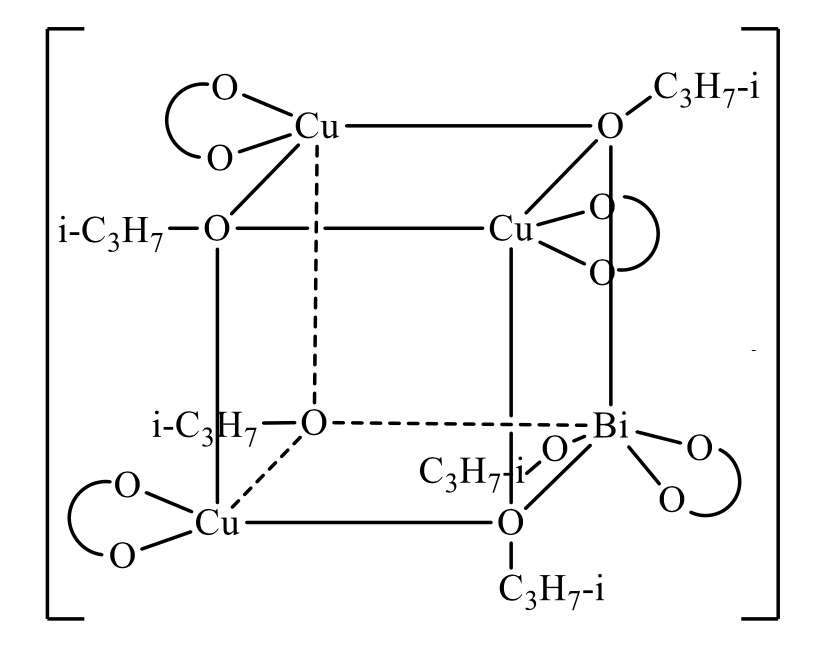
A new synthesized semiconductor material has been used as a material sensitive to the effects of temperature or magnetic field. The synthesis technique of heterometallic µ-isopropoxy-(copper(II), bismuth(III)) acetylacetonate (I) is presented in the paper. Using element analysis as well as IR-spectroscopy, magneto-chemical and thermo gravimetric researches the composition of the synthesized heterometallic µ-isopropoxy (copper(II), bismuth(III)) acetylacetonate have been examined. It corresponds to the chemical formula: [Cu3Bi(С5Н7О2)4(OC2H5)5], whereС5Н7О2 = H3C–C(O)–CH–C(O-)–CH3. The practical output is 82 % of the theoretical calculated one. The synthesized complex compound (I) is a fine crystalline powder, which has good solubility in a mixture of DMFA with chloroform (1:1) and worse solubility in alcohols, ether, better solubility in DMFA, and can be destroyed in water. An analysis of the experimental data from physical and chemical studies for compound (I) allowed to propose a chemical bonding scheme. The molecule of heterometallic µ-izopropoxy- (cuprum (II), bismuth (III)) acetylacetonate probably is a cube, and at the top of which there are three copper atoms (II) and one atom bismuth (III) linked to the atoms of oxygen of isopropoxy- groups, which acts as the tridentate ligand. For the isolated complex compound [Cu3Bi(C5H7O2)4(OC3H7-i)5], a molar mass equal to 1090.5 g/mol and a calculated number of valence electrons in one molecule is 289. A cylindrical sample of mass 0.14 g and a volume of 17.67∙10-9 m3 made of complex compound (I) by pressing was used for experimental studies. Experimental studies of the conductive properties of µ-isopropoxy- copper (II), bismuth(III)) acetylacetonate in the temperature range 313 K÷413 K, in compressed form, showed that at increased temperature its resistivity sharply decreases from 7·1010 to 4·102 Ohm∙cm. The operating temperature range is from +273 K to +493 K, with the decomposition of the chemical compound from 523 K. The concentration of charge carriers increases from 1.3·1019 m-3 at 273 K to 3.395·1036 m-3 at 493 K, while at the quantum Hall constant with increasing temperature from 273 K to 493 K decreases from 0.566 m3·Kl-1 to 2.167·10-18m3·Kl-1. The Hall voltage in the magnetic field range from 0 to 1000 mT varies from 8.32·10-14 to 8.32·10-12 V.